Moving beyond unstructured text
As early as the 2000s, it was clear that the FDA needed better tools to promote a more agile and flexible pharmaceutical manufacturing sector that could produce high quality drugs reliably with lessened regulatory oversight. For this purpose, they launched several programs and initiatives such as Process Analytical Technology (PAT), Current Good Manufacturing Practices (CGMPs), or Quality by Design (QbD), among many others. But something was still missing, as some issues continued to flow through the system: the lack of an efficient, consistent and objective process for pharmaceutical quality.
What is KASA and why it matters
In 2016, the FDA shared with the world the first prototype of the KASA System (Knowledge-Aided and Structured Application). They created the KASA system in response to increasing pressures coming from the pharmaceutical industry, public demand, and technology development. KASA uses structured data and structured information to achieve consistent and efficient regulatory reviews.
The system’s scope was to promote issue-based quality assessment using structured data and structured information to achieve consistent and efficient regulatory reviews. Before the development of KASA, due to the highly narrative nature (based on unstructured text) of the documents, it was difficult for the FDA to perform quick, comparative risk and quality checks across drug products and facilities. But all of this changed with the introduction of FDA’s new approach.
How traditional unstructured text falls short
Before the development of KASA, the FDA was burdened with an ever-growing volume of submissions, increasingly complex products and a narrative-based approach. FDA reviewers were encountering challenges such as:
- Historical data about similar products was hard to find.
- Up-to-date information was not available in a timely manner.
- Elements of the quality assessment related to risk and evaluation control approaches couldn’t be easily identified as they were written in unstructured text.
- Teams couldn’t easily manage knowledge and critical information across a product’s lifecycle.
Going one step deeper, FDA identified the following elements as the core issues that created the challenges above:
Regulatory assessments traditionally based on freestyle narratives, unstructured text, and the summarization of application information are done by cutting and pasting data and tables.
Outdated technology and existing tools cannot be used to maximize efficiency.
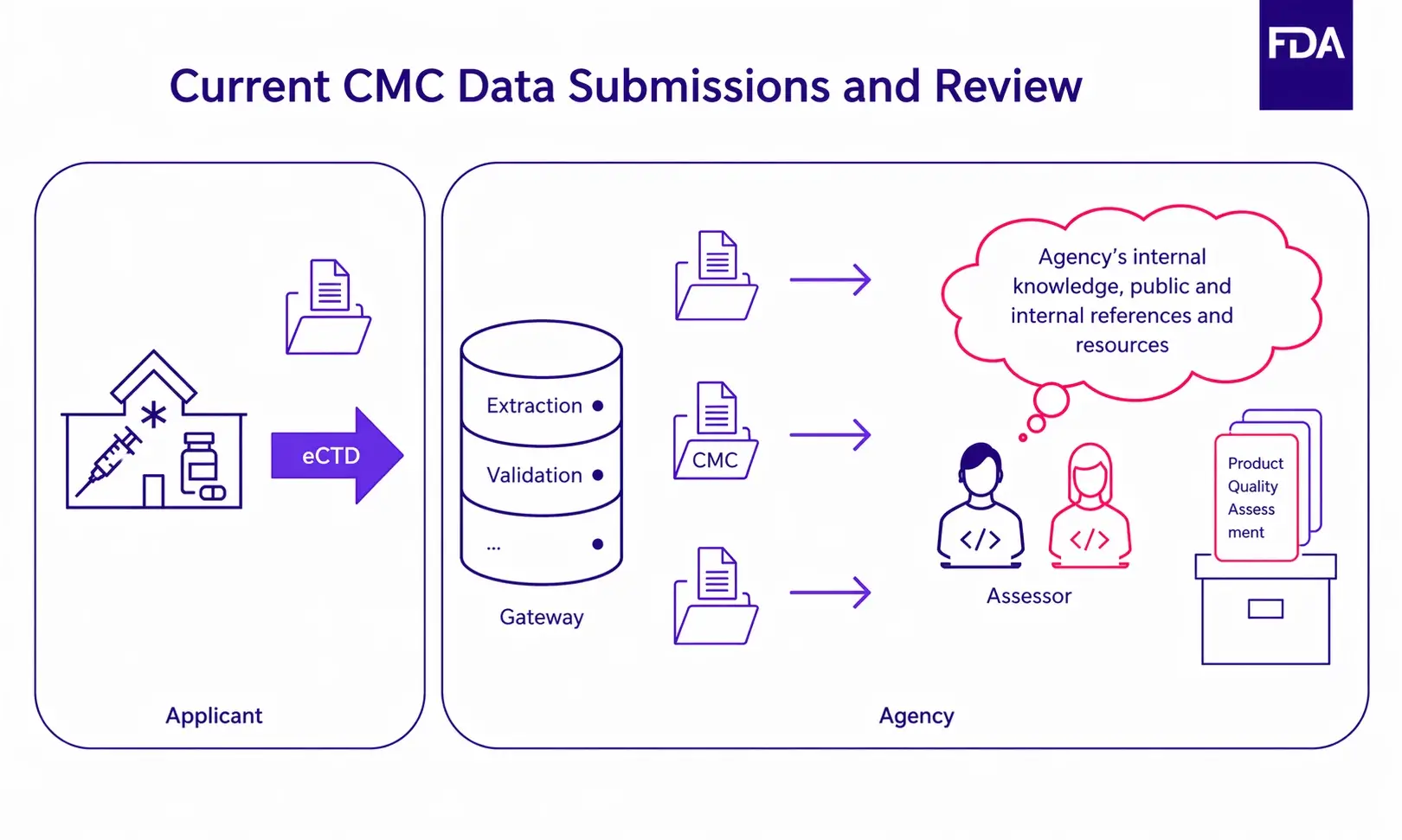
Example of a CMC data submission and review process carried out by the FDA in January 2023, before KASA
Source: FDA’s KASA and PQ/CMC Initiatives: Perspectives for Biotechnology Products
The conclusion was obvious: ineffective knowledge management tools coupled with unstructured text led to difficulties when comparing products and processes, which in the end could result in drug shortages or delays in product reviews. They needed to find a solution fast.
What is KASA?
KASA is a system that captures information about risk and control approaches for product design, manufacturing, and facility in a structured format. The shift to structured content is meant to replace freestyle, unstructured content and provide a more consistent quality assessment process for regulatory reviewers. Let’s dive deeper into what the newly proposed system entails.
FDA’s conceptual representation of KASA is shown below. The house represents KASA. The foundation represents all the historic information the agency holds on drugs and their manufacturing process.
On the left-hand side, we can see the internal part of the KASA system. The foundation of the house represents the foundation upon which reviewers rely on to assess documentation. This is the knowledge base containing all the data and information they have historically built on products from the submissions they received. Next, the pillars of the house represent the three main functions the reviewers need to perform based on the information they have. Finally, the house itself represents the Knowledge-aided Assessment activity performed by the assessors.
On the right-hand side, we can notice the input that is going into the system – the regulatory dossier that a sponsor submits. Opposed to traditional methods, this time around the dossier is represented by a structured application format based on structured content and data. Two separate but important initiatives for achieving the vision proposed through KASA are ICH M4Q and PQ-CMC, which we are going to briefly present next. They are important for further understanding the complex efforts the industry is sustaining to enable structured content applications.
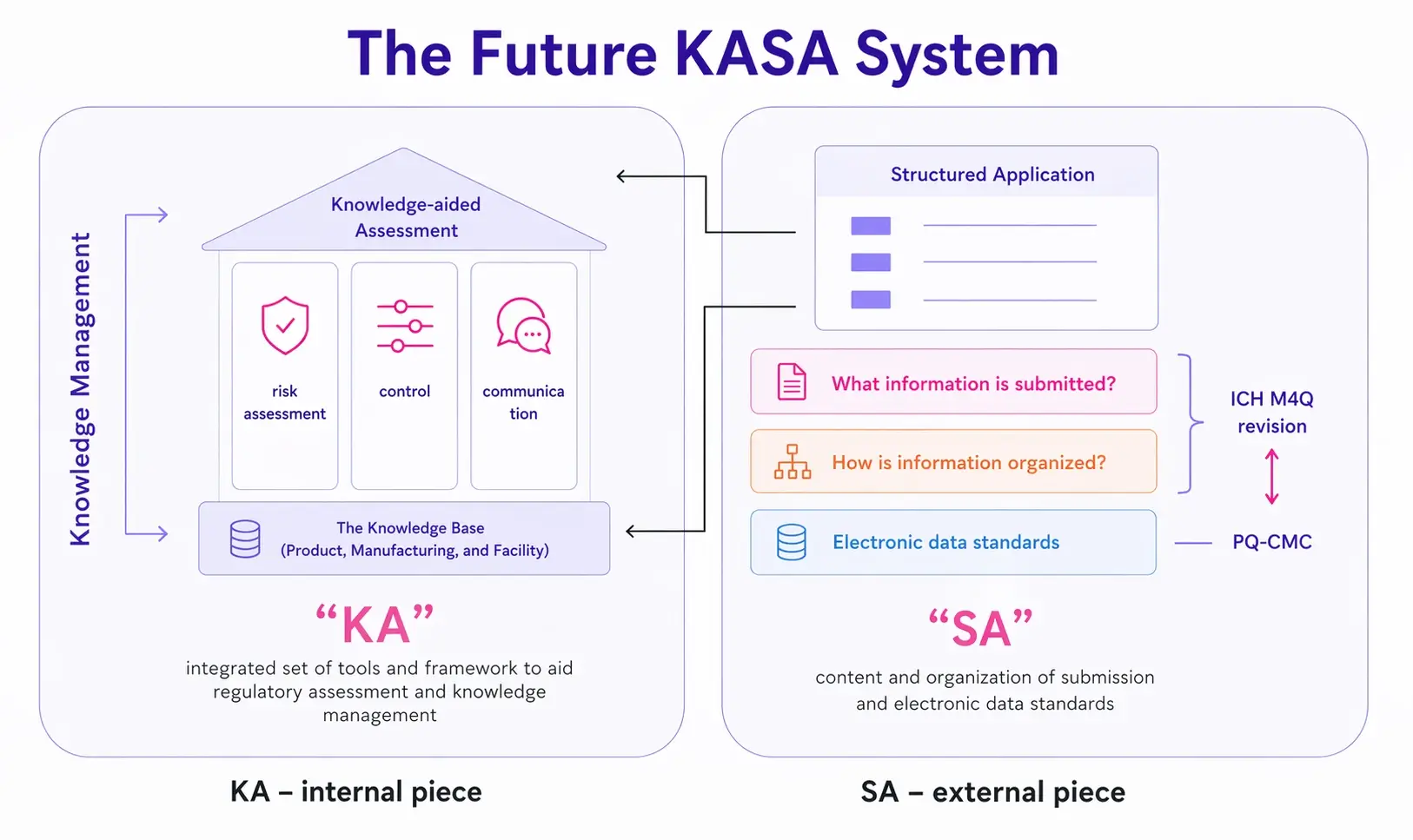
Source: FDA
ICH M4Q and PQ-CMC - two initiatives that support the implementation of KASA
ICH M4Q is a guideline within the International Council for Harmonization (ICH) framework that defines the structure and content for the "Quality" section of the Common Technical Document (CTD) used in pharmaceutical registration submissions. M4Q specifies how to organize and present information related to drug substance and drug product quality, including manufacturing processes, specifications, analytical methods, stability data and control strategies.
The main goals of ICH M4Q are to:
- Provide recommendations to sponsors on how to prepare the quality section of a drug application (Modules 2 and 3)
- Harmonize the format and organization of quality-related information across different regions (EU, US, Japan), making submission and review more efficient and consistent
- Support modern regulatory processes through standardized data, digital tools and the integration of new technologies
- Facilitate a structured regulatory assessment in FDA’s KASA system
PQ-CMC refers to Product Quality and Chemistry, Manufacturing, and Controls information submitted to the FDA. This documentation covers the scientific and regulatory framework for ensuring that pharmaceutical products are designed, manufactured and controlled to consistently meet quality standards and regulatory requirements throughout their lifecycle, covering:
- Manufacturing processes, process validation and adherence to Good Manufacturing Practice (GMP)
- Documentation of all procedures, changes, and controls for regulatory submissions and ongoing compliance
- Establishes electronic standards (data elements and terminologies) that will facilitate the transition towards a streamlined electronic review environment for the FDA
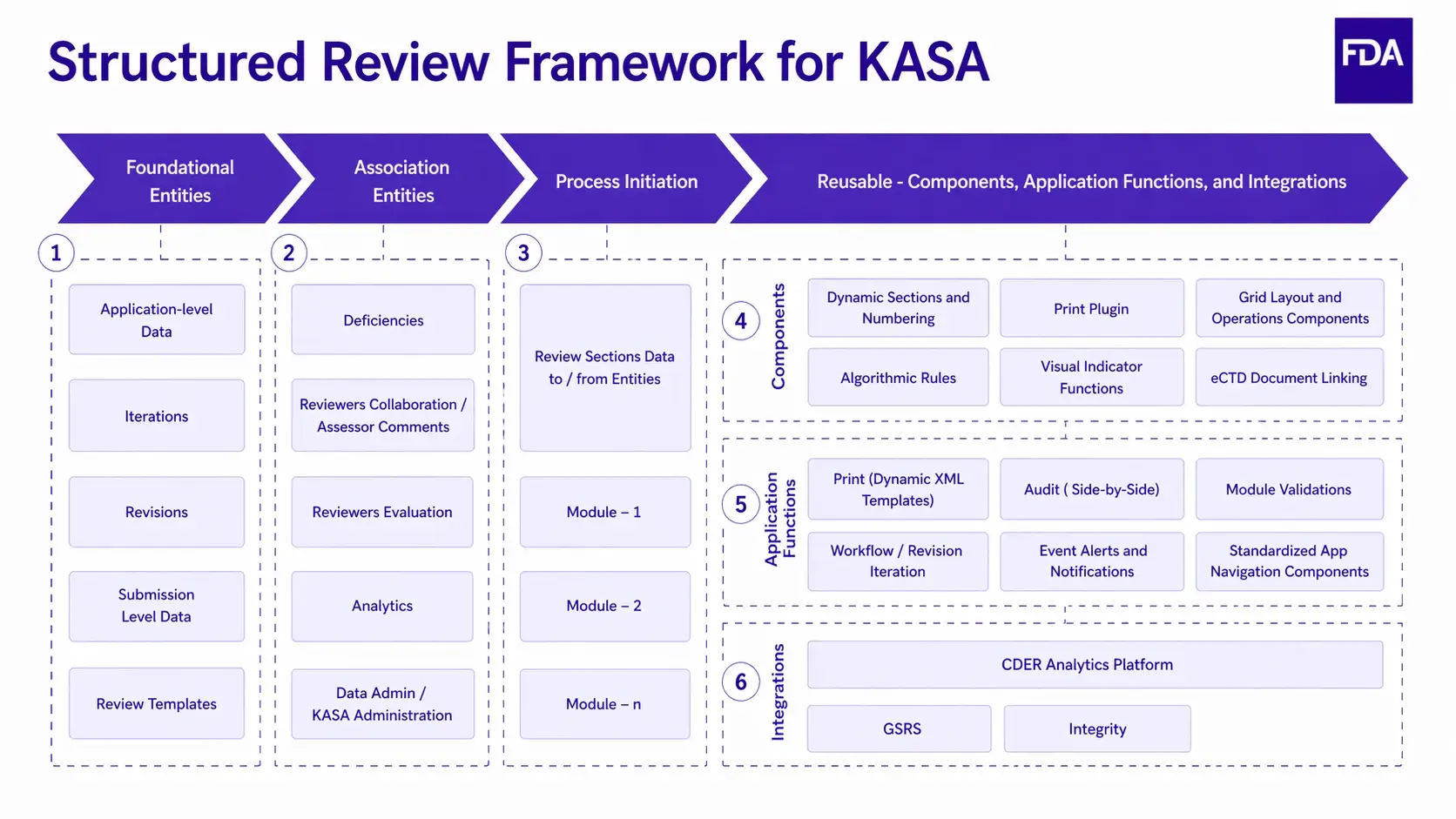
Source: Digital transformation Symposium 2023 - KASA
Above you can see the Structured Review Framework for KASA and the central role that reusable content components play in enabling the entire workflow.
According to the FDA, KASA is built to:
- Capture and manage knowledge during the lifecycle of a drug product
- Include established rules and algorithms to measure the risk associated with how a product is designed and manufactured
- Perform computer-aided analyses of applications for a comparison of regulatory standards and quality risk across the repository of approved drug products and facilities
- Provide a structured assessment that radically eliminates text-based narratives and summarization of information from the applications
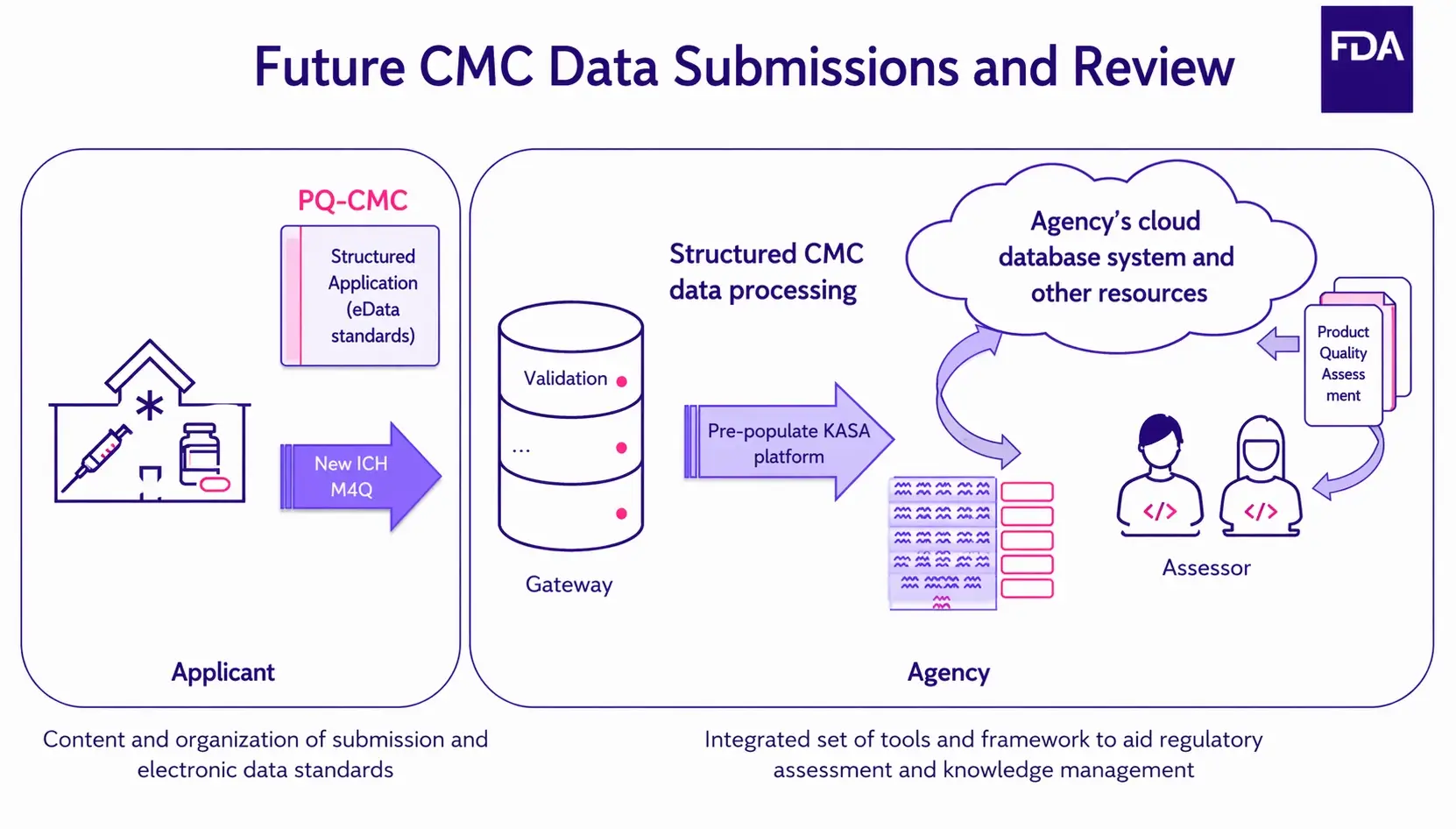
Moving away from unstructured to structured content
We have seen that KASA brings a new paradigm in terms of managing content. The initiative aims to transit all stakeholders away from unstructured content to structured data and structured information. What do these terms mean? Let’s explore.
What is Structured Content Authoring (SCA)?
With an SCA tool, sponsors can create structured regulatory submissions. They can submit regulatory documentation in any format that supports a data-centric approach, such as XML, FHIR, JSON, or other formats. This way, reviewers can receive regulatory submissions that follow the eCTD provisions stipulated in ICH M4Q, while adhering to the electronic data standards described in PQ-CMC.
An SCA solution enables sponsors to create dossiers that streamline the review process as it is an authoring method based on defining and using reusable content components. For example, information on manufacturing processes, specifications, analytical methods, and quality controls can be authored as modular blocks of content and reused across multiple submissions or product variations, as and when needed. This way of authoring represents a paradigm shift from the old ways of writing documents. And thanks to current technological developments, the experience feels and looks like writing in your familiar word processor.
Documents vs content components

Paradigm change from documents to content components
Documents vs Content components
Monolithic < > Granular, modular, reusable
Content tied to formatting < > Content separate from formatting
Lack of descriptive data < > Rich metadata, driving intelligent (re) use
Copy/paste quality issues < > Fully managed and tracked for compliance
Fixed output format < > Any output format, incl. "traditional" documents
Change management is also simplified when content is presented in chunks as opposed to monolithic documents. Updates to a single module automatically propagate to every instance where that module is referenced, supporting global consistency and lifecycle integrity. This is possible due to a mechanism called content reuse.
What is structured content reuse?
As a key aspect of structured content authoring (SCA), structured content reuse enables you to re-use any of the vetted content components anywhere you need, without having to copy/paste or otherwise duplicate it.
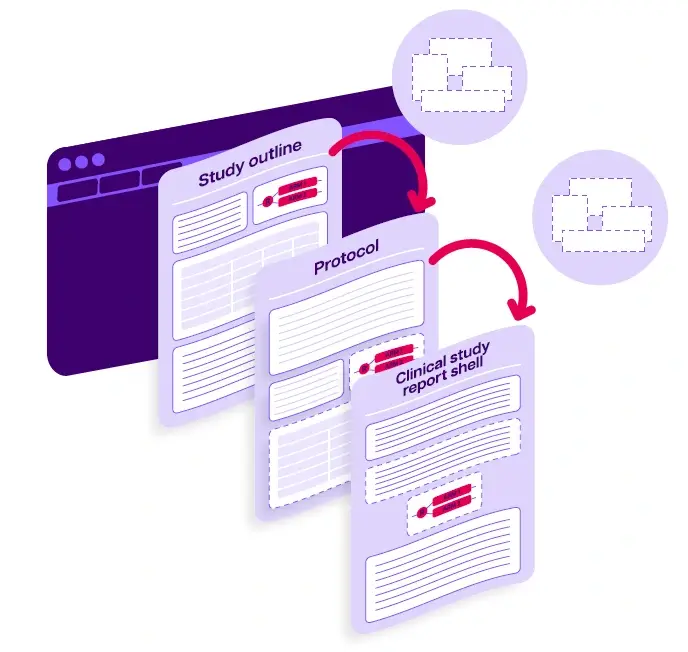
The new world: automated content reuse with AI and SCA
Filled border (–), de novo component;
Dotted border (...), reused component.
An example of content reuse between study outline, protocols, and clinical study report shells
Source: EMWA | Volume 32 number 3 | Medical writing | September 2023 | 33
Unlike outdated word processing tools, where content is monolithic and static, a structured content tool treats every table, section, paragraph, sentence, image or other elements as an independent, versioned content component. This reduces manual labor and eliminates documentation errors (or content “drift”) stemming from content duplication and inconsistent versioning.
The technology that lets you create and manage structured content components and reuse them is called a component content management system (CCMS), also known as a structured content management system (SCMS), or a Structured Content and Data Management System (SCDM). It securely stores and manages all templates, components, outputs, associated metadata, language versions, and underlying data structures.
Structured content and AI for KASA compliance
If we’ve caught your interest, keep a close eye on our blog and socials to see the next part of our article, where we are going to discuss how you can reuse content and apply AI to enable faster regulatory reviews and approvals.
In case you don’t want to wait any longer and would like to understand how you can optimize your regulatory submissions process, we are one click away.

Author
Todd Georgieff
Industry Lead for Pharma
